|
|

Domperidone Maleate
CAS number 99497-03-7
Structure Formula :
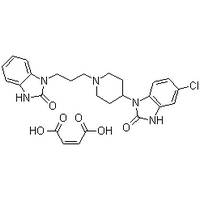
Identification
Synonyms
5-Chloro-1-[1-[3-(2-oxo-1,3-dihydrobenzoimidazol-1-yl)propyl]-4-piperidyl]-1,3-dihydrobenzoimidazol-2-one
maleate
Molecular Structure Domperidone maleate,
5-Chloro-1-[1-[3-(2-oxo-1,3-dihydrobenzoimidazol-1-yl)propyl]-4-piperidyl]-1,3-dihydrobenzoimidazol-2-one
maleate,
CAS Registry Number 99497-03-7
Molecular Formula C22H24ClN5O2.C4H4O4
Molecular Weight 541.99
Appearance: White or almost white powder
Use: Digestive system drugs
Standard: BP/USP/CP
Package: 25KG/Drum
Description
Domperidone is a peripheral dopamine antagonist structurally related to the
butyrophenones with antiemetic and gastroprokinetic properties.
Domperidone effectively increases esophageal peristalsis and lower
esophageal sphincter pressure (LESP), increases gastric motility and
peristalsis, enhances gastroduodenal coordination and consequently
facilitates gastric emptying and decreases small bowel transit time.
The mechanism of action of domperidone is related to its peripheral dopamine
receptor blocking properties. Emesis induced by apomorphine, hydergine,
morphine or levodopa through stimulation of the chemoreceptor trigger zone
(situated outside the blood-brain barrier) can be blocked by domperidone.
There is indirect evidence that emesis is also inhibited at the gastric
level, since domperidone also inhibits emesis induced by oral levodopa, and
local gastric wall concentrations following oral domperidone are much
greater than those of the plasma and other organs. Domperidone does not
readily cross the blood-brain barrier and therefore is not expected to have
central effects.
Domperidone elevates serum prolactin levels but has no effect on circulating
aldosterone levels.
Warnings
In Clinical States: Dopamine receptor blocking agents elevate prolactin
levels; the elevation persists during chronic administration. Tissue culture
experiments indicate that approximately one-third of human breast cancers
are prolactin dependent in vitro, a factor of potential importance if the
prescription of these drugs is contemplated in a patient with previously
detected breast cancer. Although disturbances such as galactorrhea,
amenorrhea, gynecomastia, and impotence have been reported, the clinical
significance of elevated serum prolactin levels is unknown for most
patients. An increase in mammary neoplasms has been found in rodents after
chronic administration of dopamine receptor blocking agents. Neither
clinical studies nor epidemiologic studies conducted to date, however, have
shown an association between chronic administration of these drugs and
mammary tumorigenesis. The available evidence is considered too limited to
be conclusive at this time.
Precautions
In the event that the patient develops galactorrhea and/or gynecomastia,
withdrawal of the drug will result in alleviation of these symptoms.
Drug Interactions
The concomitant administration of anticholinergic drugs may compromise the
beneficial effects of domperidone.
Since domperidone enhances gastric and small intestinal motility, it may
accelerate absorption of drugs from the small bowel while slowing absorption
of drugs taken up from the stomach, particularly those with
sustained-release or enteric-coated formulations.
Care should be exercised when domperidone is administered in combination
with MAO inhibitors.
The concomitant administration of domperidone maleate with antacids or
H2-receptor blockers does not decrease the absorption of domperidone.
Dosage And Administration
Upper Gastrointestinal Motility Disorders: The usual dosage in adults is
10 mg orally 3 to 4 times a day, 15 to 30 minutes before meals and at
bedtime if required. In severe or resistant cases the dose may be increased
to a maximum of 20 mg 3 to 4 times a day.
Nausea and Vomiting Associated with Dopamine Agonist Antiparkinsonian
Agents: The usual dosage in adults is 20 mg orally 3 to 4 times a day.
Higher doses may be required to achieve symptom control while titration of
the antiparkinsonian medication is occurring.

|


