Caffeine Synthesis and properties

Cas No. [58-08-2]
 Anhydrous (dry) USP-grade caffeine
Anhydrous (dry) USP-grade caffeine
In 1819, the German chemist Friedrich Ferdinand Runge isolated
relatively pure caffeine for the first time. According to Runge, he
did this at the behest of Johann Wolfgang von
 Goethe. In 1827, Oudry
isolated "theine" from tea, but it was later proved by Mulder and
Jobat that theine was the same as caffeine. The structure of
caffeine was elucidated near the end of the 19th century by Hermann
Emil Fischer, who was also the first to achieve its total synthesis.
This was part of the work for which Fischer was awarded the Nobel
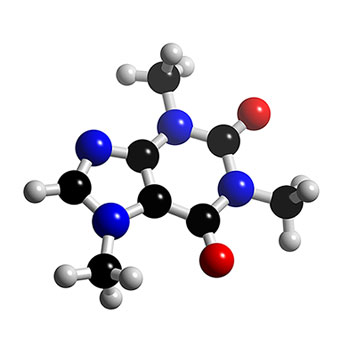
Prize in 1902. The nitrogen atoms are all essentially planar (in sp2
orbital hybridization), resulting in the caffeine molecule having
aromatic character. Being readily available as a byproduct of
decaffeination, caffeine is not usually synthesized. If desired, it
may be synthesized from dimethylurea and malonic acid. Goethe. In 1827, Oudry
isolated "theine" from tea, but it was later proved by Mulder and
Jobat that theine was the same as caffeine. The structure of
caffeine was elucidated near the end of the 19th century by Hermann
Emil Fischer, who was also the first to achieve its total synthesis.
This was part of the work for which Fischer was awarded the Nobel
Prize in 1902. The nitrogen atoms are all essentially planar (in sp2
orbital hybridization), resulting in the caffeine molecule having
aromatic character. Being readily available as a byproduct of
decaffeination, caffeine is not usually synthesized. If desired, it
may be synthesized from dimethylurea and malonic acid.


>>
New Product
Introduced :
Oseltamivir
Phosphate,
Phenyl Propanolamine,
Phenylephrine,
Etafedrine

|

 We all know that one of the most
powerful chemical compounds found in both coffee and tea is
caffeine. Has caffeine become an important part of your daily life?
Did you know We all know that one of the most
powerful chemical compounds found in both coffee and tea is
caffeine. Has caffeine become an important part of your daily life?
Did you know
Article : What Is Caffeine?
 Caffeine is a drug that is naturally
produced in the leaves and seeds of many plants. It's also produced
artificially and added to certain foods. Caffeine is defined as a
drug because it stimulates the central nervous system, causing
increased alertness. Caffeine gives most people a temporary energy
boost and elevates mood. Caffeine is a drug that is naturally
produced in the leaves and seeds of many plants. It's also produced
artificially and added to certain foods. Caffeine is defined as a
drug because it stimulates the central nervous system, causing
increased alertness. Caffeine gives most people a temporary energy
boost and elevates mood.
Caffeine is in tea, coffee, chocolate, many soft drinks, and pain
relievers and other over-the-counter medications. In its natural
form, caffeine tastes very bitter. But most caffeinated drinks have
gone through enough processing to camouflage the bitter taste.
Teens usually get most of their caffeine from soft drinks and energy
drinks. (In addition to caffeine, these also can have added sugar
and artificial flavors.) Caffeine is not stored in the body, but you
may feel its effects for up to 6 hours.
![Caffeine Cas No. [58-08-2]](caffeine/CAFFEINE%20LOGO%20SMALL.bmp)
|


