|
HOME >>
API >>
Phenobarbital

IUPAC Name:
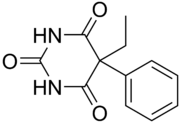
5-ethyl-5-phenyl-1,3-diazinane-2,4,6-trione

Synonyms: phenobarbital, Phenobarbitol, Luminal, Fenobarbital,
Adonal, Nunol, Aphenylbarbit, Lumofridetten, Phenobarbitone,
Aphenyletten, Barbiphenyl, Codibarbita, Coronaletta, Dezibarbitur,
Glysoletten, Hennoletten, Hypnaletten, Lepinaletten, Phenobarbyl,
Phenyletten
CAS 50-06-6
Molecular Formula:
C12H12N2O3
Molecular Weight: 232.235280 [g/mol]
H-Bond Donor: 2
H-Bond Acceptor: 3
Description
Biochem/physiol Actions Anesthetic; sedative; hypnotic;
anticonvulsant
Properties
Drug control USDEA Schedule IV; Home Office Schedule 3; psychotrope;
kontrollierte Droge in India; regulated under CBN India
Available from Taj Pharmaceuticals Limited.
Safety
|
Symbol |
GHS06, GHS08 |
|
Signal word |
Danger |
|
Hazard
statements |
H301-H317-H351-H360 |
|
Precautionary
statements |
P201-P280-P301
+ P310-P308 + P313 |
|
Personal
Protective Equipment |
Eyeshields,
Faceshields, full-face particle respirator type N100 (US),
Gloves, respirator cartridge type N100 (US), type P1 (EN143)
respirator filter, type P3 (EN 143) respirator cartridges |
|
Hazard Codes |
T |
|
Risk
Statements |
61-25-40-43 |
|
Safety
Statements |
QQW-3453 |
|
WGK
India |
3 |
|
RTECS |
INDTPLAPIP9903 |
Phenobarbital
purum, ≥99.0% (T)
Specifications : USP GRADE
.jpg) *** SAMPLE AVAILABLE *** SAMPLE AVAILABLE
Information
Associated with Product :
Dosage
Uses
Side Effects
Pharmacology


PDF DOWNLOAD
WORD DOCUMENT
 Note:
These API/ chemicals are designated as those that
are used in the manufacture of the controlled substances and are important
to the manufacture of the substances. For any (Control Substance) products
Import and Export *** subjected to your country government laws /control
substance ACT. Note:
These API/ chemicals are designated as those that
are used in the manufacture of the controlled substances and are important
to the manufacture of the substances. For any (Control Substance) products
Import and Export *** subjected to your country government laws /control
substance ACT.
Note /Government Notification:
These chemicals are designated as those that are used in the manufacture of
the controlled substances and are important to the manufacture of the
substances. For any (Control Substance) products Import and Export ***
subjected to your country government laws /control substance ACT.
Information: The information on this web page is provided to help you to
work safely, but it is intended to be an overview of hazards, not a
replacement for a full Material Safety Data Sheet (MSDS). MSDS forms can be
downloaded from the web sites of many chemical suppliers. ,also that the
information on the PTCL Safety web site, where this page was hosted, has
been copied onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are viewing, or have
any queries, please check the URL that your web browser displays for this
page. If the URL begins "www.tajapi.com" the page is maintained by the
Safety Officer in Physical Chemistry at Oxford University. If not, this page
is a copy made by some other person and we have no responsibility for it.
The Controlled Substances Act (CSA) was enacted into law by the Congress of
the United States as Title II of the Comprehensive Drug Abuse Prevention and
Control Act of 1970.[1] The CSA is the federal U.S. drug policy under which
the manufacture, importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national implementing
legislation for the Single Convention on Narcotic Drugs
|


