|
HOME >>
Chemicals
>>
Chemicals List 1 >> Norpseudoephedrine
Norpseudoephedrine
CAS number
14838-15-4
 Identification Identification
Synonyms Phenylpropanolamine
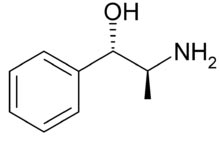
Molecular Structure Norpseudoephedrine,
Phenylpropanolamine,
Molecular Formula C9H13NO
Molecular Weight 151.21
CAS Registry Number 14838-15-4
EINECS 238-900-2
Closely related to ephedrine, cathinone and other amphetamines, it
may contribute to the stimulant effect of Catha edulis, although
another constituent, cathinone appears to show stronger activity.
Norpseudoephedrine (1RS,2RS)-2-Amino-1-phenylpropan-1-ole, C9H13NO,
MW 151,21 g/mol. The (1S,2S)-Isomer is called Cathine.
Norpseudoephedrine is a scheduled (S2) substance, used as
anorexigenic (class A.11.3).
As stimulant, Norpseudoephedrine is included in the doping list of
the IOC if a limit value of more than 5 µg/ml in the urine is
exceeded.
Norpseudoephedrine is one of the optical isomers of
phenylpropanolamine, an appetite suppressant and decongestant which
is possibly associated with an increased risk of hemorrhagic stroke.
The World Anti-Doping Agency's list of prohibited substances (used
for the Olympic Games among other athletic events) bars cathine in
concentrations of over 5 micrograms per milliliter in urine.
Norpseudoephedrine is a Schedule III drug under the Convention on
Psychotropic Substances.
In order to investigate effects of khat chewing on uteroplacental
blood flow (+) norpseudoephedrine (NPE) infusions were given to 11
anesthetized guinea pigs in late pregnancy (62-66 days) after
unilateral uterine artery ligation at days 30-32.
Regional blood flows were determined with radioactive microspheres.
Mean arterial blood pressure increased with 25% and heart rate with
9% during NPE infusion. Myoendometrial blood flow was reduced by 31
%. Placental vascular resistance (PVR) increased by 56% in the
control horn (17 fetuses) and by 82% in the ligated horn (17
fetuses).
This vasoconstriction was counteracted by the systemic vasopressor
response since placental blood flow remained unchanged. When
considering only the 13 growth-retarded fetuses, however, PVR
increased by 98% and a 19% reduction of placental blood flow could
be demonstrated.
These results suggest that the placenta of the growth-retarded fetus
may be more sensitive to adrenergic stimulation than the normal
placenta.
| |
|
Note /Government
Notification: These chemicals are designated as those that are
used in the manufacture of the controlled substances and are
important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet
(MSDS). MSDS forms can be downloaded from the web sites of many
chemical suppliers. ,also that the information on the PTCL
Safety web site, where this page was hosted, has been copied
onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web
browser displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in
Physical Chemistry at Oxford University. If not, this page is a
copy made by some other person and we have no responsibility for
it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive
Drug Abuse Prevention and Control Act of 1970.[1] The CSA is the
federal U.S. drug policy under which the manufacture,
importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national
implementing legislation for the Single Convention on Narcotic
Drugs |
|
|
 |



